
Boiling point of water |
|
100 degrees Celsius |
| |

Density |
|
the amount of mass (or matter) compacted into a certain volume |
| |

Solid |
|
has a specific volume and shape and the particles are held tightly together |
| |

Freezing point of Water |
|
0 degrees Celsius |
| |

Liquid |
|
has specific volume but not a specific shape |
| |

Thermometer |
|
instrument for measuring temperature |
| |

Gas |
|
has no specific volume or shape and the particles move freely |
| |

Balance Scale |
|
the instrument for measuring mass |
| |

Mixture |
|
a combination of two or more substances |
| |

Magnet |
|
attracts iron or stel and can be used to separate a mixture of sand and iron filings |
| |

Solution |
|
a mixture of two or more substances that are evenly distributed |
| |

Insulator |
|
any material that stops or slows the movement of electricity or heat |
| |

Dissolve |
|
to completely mix together |
| |

Conductor |
|
any material that easily allows heat or electricity to move through it |
| |

Physical Property |
|
a characteristic that can be measured or observed without changing the substance |
| |

Filter |
|
a tool used to separate smaller particles from larger particles, such as separating sand and water |
| |
|
Mass |
|
the amount of matter in an object measured in grams |
| |
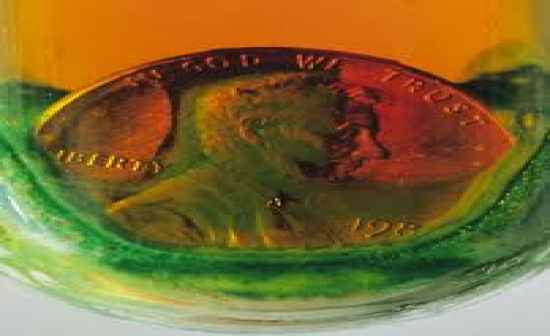
Chemical Change |
|
when two substances join and a new substance is created (ex. mixing baking soda with water) |
| |

Physical Change |
|
the substance changes size or shape, but is still the same object |
| |

Buoyancy |
|
the ability or tendency to float in water or air or some other fluid |
| |

Signs of Physical Change |
|
Change in size, shape, or state of matter (liquid to solid, solid to liquid, liquid to gas) |
| |

Signs of Chemical Change |
|
bubbles, heat given off, light given off, new substance formed, color change |
| |
